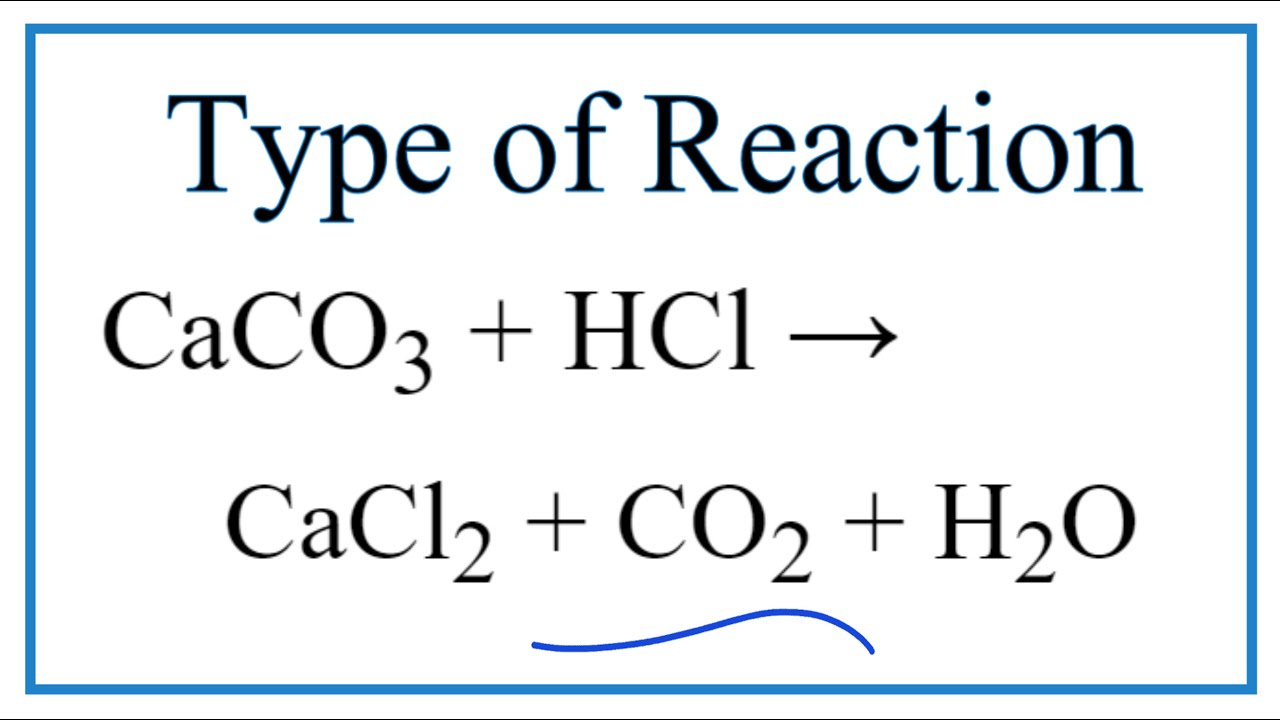
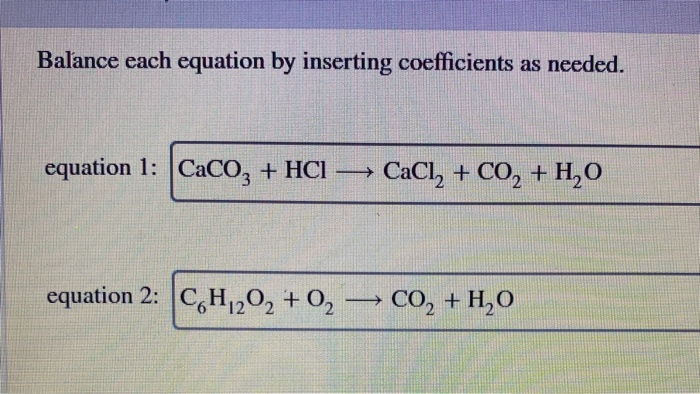
Q8. Balance the following reactions: a) CaCO3 + HCl +CaCl2 + H2O + CO2 b) H2O + H2 + O2 c) N2 + H2 → NH d) NaOH + H2SO4 → Na

HCl+CaCO3=CaCl2+H2O+CO2 balance the chemical equation @mydocumentary838. hcl +caco3=cacl2+h2o+co2 - YouTube
The mass of CaCO3 required to react completely with 20 mL of 1.0 M HCL as per the reaction CaCO3+2HCl–>CaCl2+CO2+H2O
40. Consider the reaction CaCO3+2HCL (l) 》CaCl2+CO2+H2O (l).what mass of CaCO3 is required to react with 20mL 1M HCL?
16. In a chemical reaction, caco3+2hcl= cacl2 +co2+h2o. 25ml hcl and 0.75M Calculate the amount of caco3